Donna Haraway opens When Species Meet with the proclamation that “human genomes can be found in only about 10 percent of all the cells that occupy the mundane space I call my body” (Haraway, 2007). The rest of the human body is comprised of bacteria, viruses, archaea and other microorganisms – largely invisible to the human eye, but nonetheless living on and within us. In recent years we have seen a proliferation of research and attention towards these microorganisms, particularly in the gut, skin, lungs, and mouth, with each comprising their own microbiome. Although this research is still developing, it is increasingly clear that these microorganisms may actually be fundamental to the development and functioning of human bodies.
Germs or Old Friends?
Since the emergence of germ theory in the 1880’s, we have largely considered microorganisms as pathogenic, harmful agents that cause disease if allowed to enter the body (Cohen, 2009). Since then, our antimicrobial efforts have significantly reduced rates of infectious disease through increased sanitation, antibiotics, anti-parasite programs, food safety guidelines and the treatment of drinking water. However, with new research finding that the microbiome plays a role in many bodily processes that affect health, many people are now wondering if our reduced exposure to microbes may actually have unintended consequences.
The ‘old friends’ hypothesis argues that many microbes found in the human microbiome are beneficial, or at least benign, and that early and regular exposure to diverse microbial environments is critical for the development and ‘calibration’ of the immune system (Rook, 2009). Certain microbes, sometimes called ‘old friends,’ have co-evolved with humans, and over time have trained or modulated the human immune system to accept their presence. These microbes play an active role in the immune system, modulating immune response in a beneficial way. Modern lifestyles, cleanliness and sanitation, which have all reduced our microbial biodiversity, may be contributing to rising rates of chronic illnesses, including allergies and autoimmune disease (Parker et al., 2012). Dysbiosis of certain microbes in the gut are now hypothesised to impact anxiety, depression and other mental health conditions, through what is termed the ‘gut–brain axis’ (Xiong et al., 2023).
The Body as an Ecosystem
The term holobiont was first introduced by evolutionary biologist Lynn Margulis, whose pioneering theory of symbiogenesis, literally ‘becoming by living together’ argued that symbiosis, or the interactions between organisms, is a key driver of evolutionary processes (Margulis & Fester, 1991). A ‘holobiont’ is a symbiotic assemblage of different species that together form an ecological unit (Benezra et al., 2012). What might it mean to reconceptualise the human body and its microbiome as a holobiont? In this view, health isn’t just about your body—it’s about how well that whole ecosystem works together.
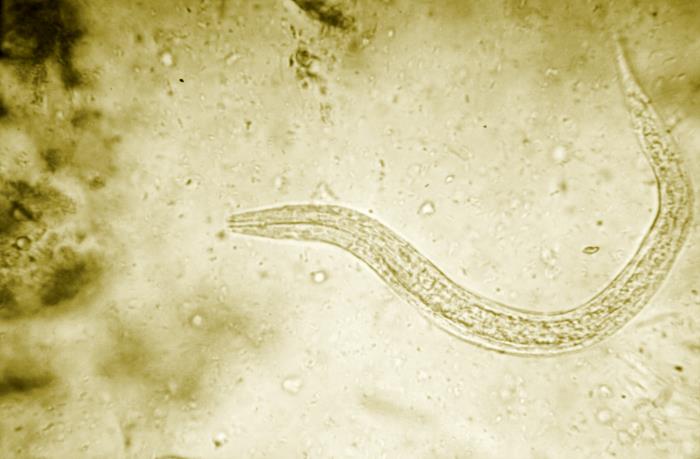
Figure 1. Hookworm larva. Image from Public Health Image Library, Centers for Disease Control and Prevention.
One surprising example comes from research on helminths, or parasitic worms. These worms were once common in human guts but have largely disappeared in industrialised countries due to sanitation and deworming campaigns. While helminths can absolutely cause harm, some researchers are now asking whether their total removal has unintended consequences, believing that in some cases, helminths may actually calm immune responses. This may be particularly important in cases of autoimmune disease, where the body attacks its own cells.
Ethnographic research by Jamie Lorimer (2016, 2019) investigated the emergence of helminthic therapy, where individuals chose to deliberately (re)introduce worms into their bodies to treat autoimmune and inflammatory conditions. Lorimer’s interlocutors “care for their worms as friends”, making continual sacrifices to their bodies, lives and routines to ensure that their bodies remain a hospitable environment to the worms (Lorimer, 2016, p. 72). In return, they hope that these ‘gut buddies’ will perform their own role in their body’s ecosystem – calming inflammation and reducing autoimmune responses. They weren’t trying to eradicate microorganisms—they were trying to live in collaboration with them.
Anthropologist Ann Kelly looks at another side of human–microbe entanglement in her work on malaria. The malaria parasite has co-evolved with both humans and mosquitoes. Despite our best efforts to ‘disentangle’ ourselves from the parasite, it has adapted relentlessly to our attempts to eradicate it, including through developing resistance to chloroquine, the drug that was previously used as a first line treatment for malaria infections. Kelly (2012) utilises the concept of ‘hospitality,’ to characterise these relationships – both between researchers and local communities in Tanzania, and also between humans, mosquitos and the malaria parasites. In coexisting in the same space, we are forced to form “relationships of (troubled) coexistence” (Kelly, 2012, p. 156).
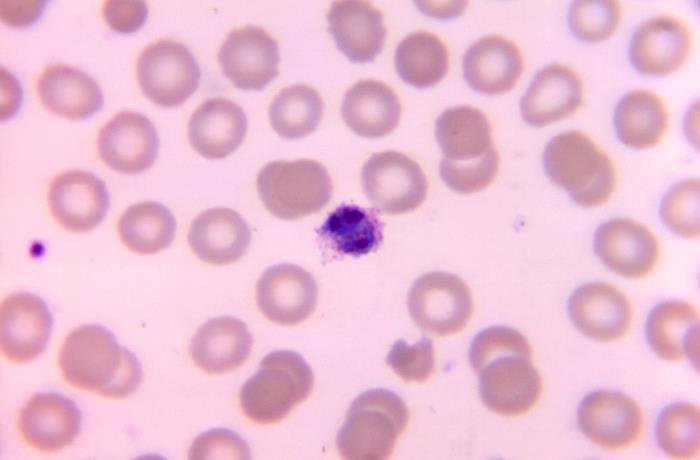
Figure 2. Micrograph of a blood smear revealing Plasmodium ovale, one of several species responsible for malaria in humans. Image from Public Health Image Library, Centers for Disease Control and Prevention.
Medical anthropologist Roberta Raffaetà similarly argues for an ecosystemic view of health that takes account of our cohabitation with the non-human. In her ethnography of microbiome research laboratories, Raffaetà (2022) traces the ways in which the narrative of symbiotic coexistence between microorganisms and human beings came to be translated into laboratory practice. It is now understood that the microbiome plays a role in many bodily processes that affect health including endocrine function, immune response, cardiovascular health, and neurodevelopment. In turn, life experiences such as diet, stress and environmental exposures all impact the microbiome. Raffaetà suggests that health is “a property not just of one body but as the emergent property of a network of relationships” (Raffaetà, 2022, p. 1). In taking this approach, health can no longer be situated only within the individual. Instead, it demands that we take account of the interaction and relationships within dynamic, mutually constituting ecosystems.
The holobiont concept offers a compelling lens for understanding this interconnectedness of living systems, within and outside the individual body. Instead, it encourages us to recognise that the body is permeable, and that we are living with and alongside the gut, skin, and other microbiomes – interconnected and always impacted by a myriad of interactions and relationships. It challenges biomedical understandings that construct the individual, physical and bounded body as the primary site of health.
The Human Holobiont
As microbiome research unsettles our assumptions about microorganisms and the role they play in human bodies, the holobiont concept provides a framework for understanding health as relational and ecological. In attending to the fact that humans are holobionts – ecosystems within which microorganisms play an active, critical role – we reconceptualise the body as an environment. In a discussion of changing notions of the immune system, Ford and Swallow (2024) argue, “if one is part of an ecology, the environment is not ‘out there’, rather, it is us. We are inextricable from what surrounds and composes us. We don’t exist without it” (p.3). This approach forces us to acknowledge the fact that environments are multi-scalar and co-constituting, troubling the distinctions between the body and its outside environment – they emerge simultaneously as they are mutually implicated.
While these types of entanglement have already been highlighted by anthropologist and feminist STS scholars, who have long troubled the boundaries between concepts of the social and biological (Franklin, 2003; Niewöhner & Lock, 2018), the holobiont emerges as a useful concept through which we can continue to de-individualise our understandings of health, and better attend to the myriad of factors and actors that impact on illness and disease. How might we manage our microbial and non-human entanglements and learn to live with, rather than eradicating, the non-human? As Haraway says, “to be one is always to become with many” (Haraway, 2007, p. 4).
This post was reviewed by Contributing Editor Shreyasha Paudel.
References
Benezra, A., DeStefano, J., & Gordon, J. I. (2012). “Anthropology of Microbes.” Proceedings of the National Academy of Sciences, 109(17), 6378–6381. https://doi.org/10.1073/pnas.1200515109
Cohen, E. (2009). A Body Worth Defending: Immunity, Biopolitics, and the Apotheosis of the Modern Body. Duke University Press. https://doi.org/10.1215/9780822391111
Ford, A., & Swallow, J. (2024). “The Immune System, Immunity and Immune Logics: Troubling Fixed Boundaries and (Re)conceptualizing Relations.” Medicine Anthropology Theory, 11(1), 1–12. https://doi.org/10.17157/mat.11.1.9443
Franklin, S. (2003). “Re-thinking Nature—Culture: Anthropology and the New Genetics.” Anthropological Theory, 3(1), 65–85. https://doi.org/10.1177/1463499603003001752
Haraway, D. (2007). When Species Meet. University of Minnesota Press.
Kelly, A. H. (2012). “The Experimental Hut: Hosting Vectors.” Journal of the Royal Anthropological Institute, 18(s1). https://doi.org/10.1111/j.1467-9655.2012.01769.x
Lorimer, J. (2016). “Gut Buddies: Multispecies Studies and the Microbiome.” Environmental Humanities, 8(1), 57–76. https://doi.org/10.1215/22011919-3527722
Lorimer, J. (2019). “Hookworms Make Us Human: The Microbiome, Eco‐immunology, and a Probiotic Turn in Western Health Care.” Medical Anthropology Quarterly, 33(1), 60–79. https://doi.org/10.1111/maq.12466
Margulis, L., & Fester, R. (Eds.). (1991). Symbiosis as a source of evolutionary innovation—Speciation and Morphogen. MIT Press Ltd.
Niewöhner, J., & Lock, M. (2018). “Situating Local Biologies: Anthropological Perspectives on Environment/Human Entanglements.” BioSocieties, 13(4), 681–697. https://doi.org/10.1057/s41292-017-0089-5
Parker, W., Perkins, S. E., Harker, M., & Muehlenbein, M. P. (2012). “A Prescription for Clinical Immunology: The Pills are Available and Ready for Testing.” Current Medical Research and Opinion, 28(7), 1193–1202. https://doi.org/10.1185/03007995.2012.695731
Raffaetà, R. (2022). Metagenomic Futures: How Microbiome Research Is Reconfiguring Health and What It Means to Be Human (1st ed.). Routledge. https://doi.org/10.4324/9781003222965
Rook, G. A. W. (2009). The Hygiene Hypothesis and Darwinian Medicine. Birkhäuser.
Xiong, R.-G., Li, J., Cheng, J., Zhou, D.-D., Wu, S.-X., Huang, S.-Y., Saimaiti, A., Yang, Z.-J., Gan, R.-Y., & Li, H.-B. (2023). “The Role of Gut Microbiota in Anxiety, Depression, and Other Mental Disorders as Well as the Protective Effects of Dietary Components.” Nutrients, 15(14), 3258. https://doi.org/10.3390/nu15143258
